|
However, once you get to Gallium, n=3 is now full, but we're not done with that row of the periodic table, so current row and unfilled shells are not equivalent anymore either. The valence shell of main-group elements is made up. The outermost shell would be n=4 only, but the current row and unfilled shells both include n=3 and n=4. As a result, valence electrons have a direct impact on the behaviour of elements during a chemical reaction. If one of my answers from before was correct, awesome, but they're no longer identical. But that would mean n=4 has electrons in it before n=3 gets filled. And the number in front should be the same thing as the rings of the Bohr model, right? The first one holds 2, then 8, 18, 32 which is the same for Schrödinger's model when n=1, 2, 3, and 4. Specifically, I know that 4s fills before 3d. 
My main question is what are valence electrons when we graduate from the Bohr model to the orbitals/Schrödinger model. Answering that would be cool, but it's not my main question. Now, I know weird things can happen if an electron gets excited and I don't know if that has an effect on which electrons are considered valence or not or if it only matters what the ground state looks like. I don't know how valence electrons were described to me back then, but I took that outermost shell, electrons that were added while walking along that row of the periodic table, and electrons that were in an unfilled shell (except for noble gases) to all be equivalent definitions. Oxygen will gain 2 electrons.I learned the Bohr model first (I'm sure like most students) and each shell had to be filled before the next shell could start. The second shell has six electrons ( 2 s 22 p 4) and needs two electrons to achieve octet. The electron configuration of O atom is 1 s 22 s 22 p 4. How many electrons must O lose/gain to achieve octet? Write the formula of the resulting ion and its electron configuration. Write the electron configuration of oxygen atom (Z=8). In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. 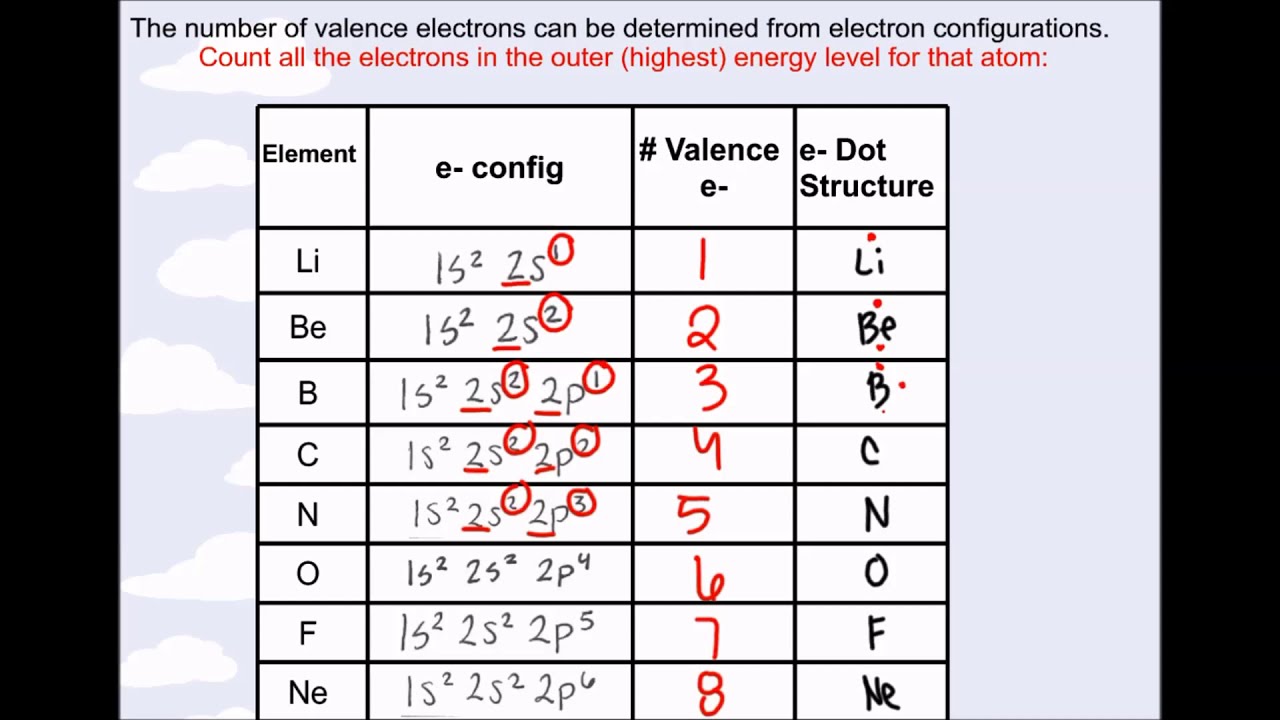
For example, the electron dot diagram for iron (valence shell configuration 4s 2 3d 6) is as follows. The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. For atoms with partially filled d or f subshells, these electrons are typically omitted from Lewis electron dot diagrams. 
Notice that there are no leftover electrons. Alkali metals are the least electronegative (most electropositive) elements in the periodic table due to a presence of a single valence electron that is donated very easily. The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. The presence of valence electrons can determine the elements. A valence electron is an electron that is associated with an atom, and that can participate in the formation of a chemical bond in a single covalent bond, both atoms in the bond contribute one valence electron in order to form a shared pair. On the left, the chlorine atom has 17 electrons. 1.3: Valence electrons and open valences.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
